The COVID-19 pandemic, caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has had a profound impact on global health. While much attention has been focused on the acute phase of the disease, there is increasing recognition of a condition known as “Long COVID” or “post-acute sequelae of SARS-CoV-2 infection” (PASC). Long COVID is characterized by a range of persistent symptoms that can last for weeks or even months after the acute infection has resolved. One of the most prevalent and distressing symptoms of Long COVID is the loss of the sense of taste and smell, a condition known as anosmia and ageusia, respectively.
Anosmia and ageusia are not unique to Long COVID; they are also common acute symptoms of COVID-19 itself. Research has suggested that these sensory deficits may be related to the neural pathways affected by the virus. Interestingly, a potential treatment avenue has emerged in the form of vagus nerve stimulation (VNS). This article explores the effects of VNS as a potential adjunct therapy for Long COVID recovery, drawing upon recent research and clinical observations.
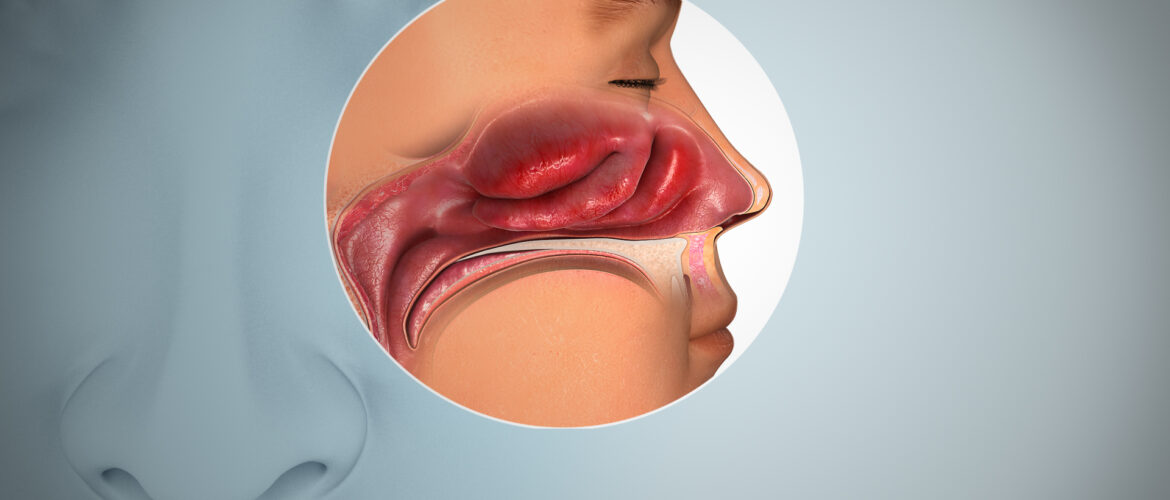
The Impact of Anosmia and Ageusia in Long COVID
Anosmia and ageusia can significantly impact an individual’s quality of life. Beyond the loss of enjoyment associated with eating and drinking, these sensory deficits can have safety implications, such as an inability to detect spoiled food or the smell of smoke in case of a fire. Moreover, these symptoms can contribute to depression, anxiety, and social isolation.
Recent studies have reported that between 20% and 85% of COVID-19 patients experience anosmia and ageusia to varying degrees. However, the pathophysiology of these symptoms in both acute and Long COVID remains incompletely understood, and effective treatments are limited.
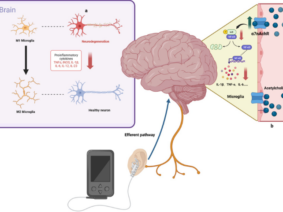
Vagus Nerve Stimulation as a Promising Therapy
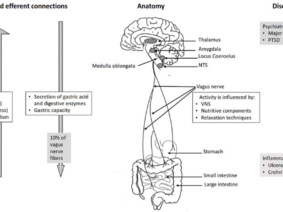
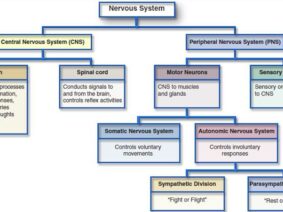
Vagus nerve stimulation is a neuromodulation technique that has shown promise in various medical applications. It involves the use of electrical impulses to stimulate the vagus nerve, which is a key component of the parasympathetic nervous system. This stimulation has been found to have anti-inflammatory and immunomodulatory effects, making it a potentially valuable tool in the context of COVID-19.
Two key studies shed light on the potential of VNS in COVID-19:
- Pehlivanoglu et al. (2023): In a case report published in “Med Records,” Pehlivanoglu and colleagues presented a 27-year-old female patient with anosmia and ageusia lasting for over a year. They applied transcutaneous ear vagus nerve stimulation daily for 15 days. The study reported a progressive improvement in the sense of taste and smell. While the results were promising for taste, the effect on smell and odor discrimination was less significant.
- Azabou et al. (2021): Azabou and his team explored the potential of VNS as an adjunct therapy for COVID-19 in an article published in “Frontiers in Medicine.” Their research suggested that VNS could attenuate inflammation in COVID-19 patients, and it was considered a non-drug therapeutic strategy. Clinical trials investigating VNS as an adjunct therapy in COVID-19 were underway, indicating a growing interest in this approach.
Long COVID presents a unique set of challenges for patients and healthcare providers. The persistent loss of taste and smell is just one of many debilitating symptoms faced by individuals in their recovery journey. Emerging research, as exemplified by the studies of Pehlivanoglu et al. and Azabou et al., suggests that Vagus Nerve Stimulation may offer a glimmer of hope in addressing this specific aspect of Long COVID. While more research and clinical trials are needed to establish its efficacy definitively, VNS stands as a promising avenue for improving the lives of those affected by Long COVID’s sensory deficits.
References:
- Pehlivanoglu BE, Guven S, Hatik SH, Ozden AV. (2023) “The Effect of Vagus Nerve Stimulation Applications on Taste and Smell Loss in COVID-19 Syndrome: Case Report.” Med Records, 5(3), 655-8. DOI: 10.37990/medr.1262924.
- Azabou E, Bao G, Bounab R, Heming N, Annane D. (2021) “Vagus Nerve Stimulation: A Potential Adjunct Therapy for COVID-19.” Frontiers in Medicine, 8, 625836. DOI: 10.3389/fmed.2021.625836.